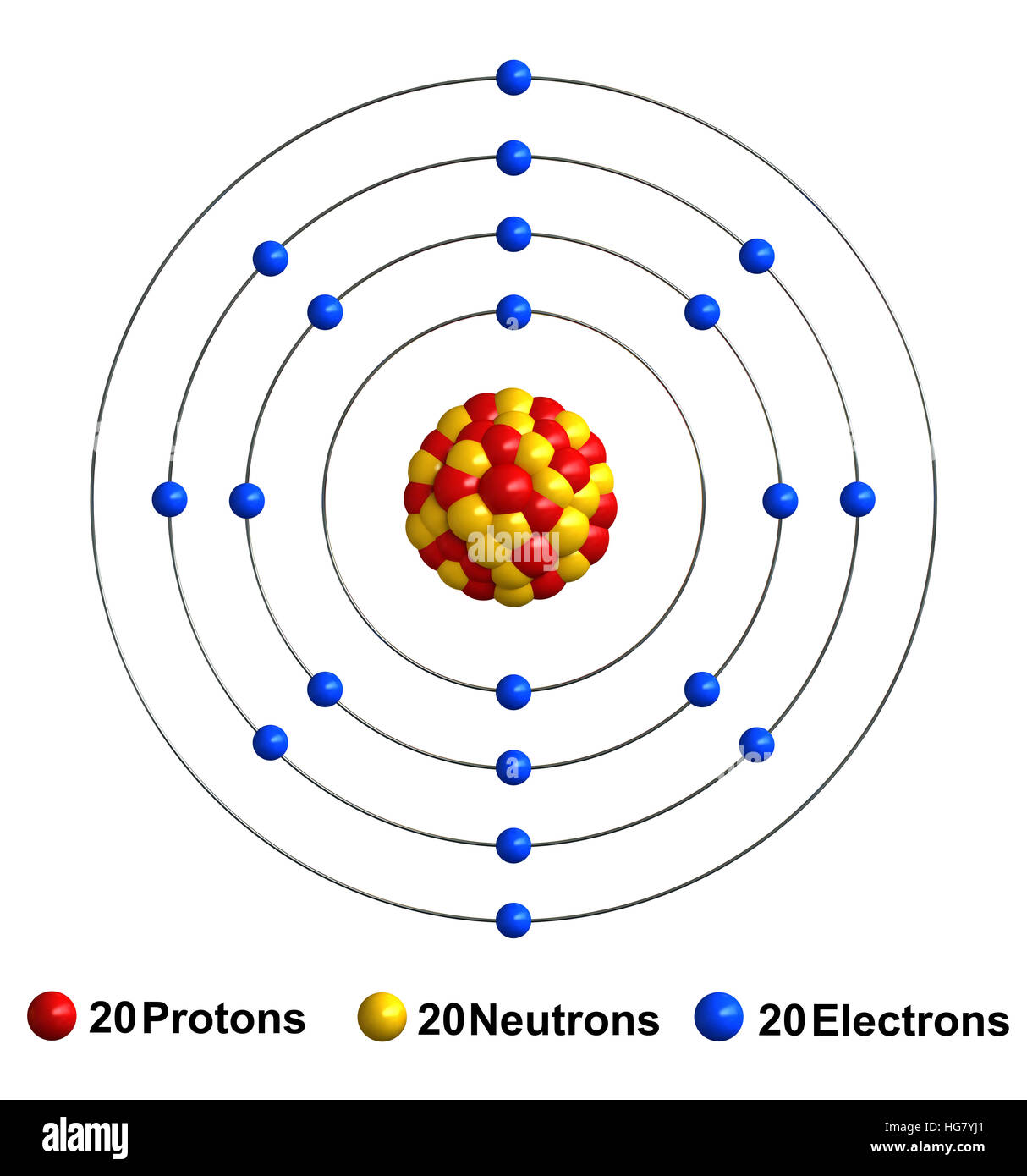
The name "calcium" originates from the Latin word "calics," meaning lime. It is also an biologically essential substance found in teeth, bones, and shells. It frequently serves as an alloying agent for other metals like aluminum and beryllium, and industrial materials like cement and mortar are composed of calcium compounds like calcium carbonate. Calcium is a reactive, soft metal that is a member of the alkaline earth elements. In its elemental form, calcium has a dull gray-silver appearance. It is the fifth most abundant element in the earth's crust and can be found in minerals such as dolomite, gypsum, plagioclases, amphiboles, pyroxenes and garnets. Calcium was discovered and first isolated by Sir Humphrey Davy in 1808. Atomic Number: 20: Atomic Mass: 40.078 atomic mass units: Number of Protons: 20: Number of Neutrons: 20: Number of Electrons: 20: Melting Point: 839. The calcium atom has a radius of 197 pm and a Van der Waals radius of 231 pm. One atomic mass unit is equal to 1.66 x 10 -24 grams. The unit of measure for mass is the atomic mass unit (amu). Therefore this resulting atomic mass is calculated from naturally-occurring isotopes and their abundance. The number of electrons in each of Calcium's shells is and its electron configuration is 4s 2. Note that each element may contain more isotopes. Calcium (atomic symbol: Ca, atomic number: 20) is a Block S, Group 2, Period 4 element with an atomic weight of 40.078. LICENSED GRANTED TO MAKE UNLIMITED PAPER COPIES FOR INTERNAL USE ONLY. See reverse side of invoice or packing slip for additional terms and conditions of sale. American Elements shall not be held liable for any damage resulting from handling or from contact with the above product. It does not represent any guarantee of the properties of the product. The information in this document is based on the present state of our knowledge and is applicable to the product with regard to appropriate safety precautions. The above information is believed to be correct but does not purport to be all inclusive and shall be used only as a guide. Safety Data Sheet according to Regulation (EC) No. Relevant identified uses of the substance: Scientific research and development It is the fifth most abundant element in Earth's crust. Its physical and chemical properties are most similar to its heavier homologues strontium and barium. As an alkaline earth metal, calcium is a reactive metal that forms a dark oxide-nitride layer when exposed to air. Product Number: All applicable American Elements product codes, e.g. Calcium is a chemical element with the symbol Ca and atomic number 20. Thin Film Deposition & Evaporation Materials.Additive Manufacturing & 3D Printing Materials.Thus, the columns of the periodic table represent the potential shared state of these elements' outer electron shells that is responsible for their similar chemical characteristics. When an atom gains an electron to become a negatively-charged ion this is indicated by a minus sign after the element symbol for example, \(F^-\). Group 17 elements, including fluorine and chlorine, have seven electrons in their outermost shells they tend to fill this shell by gaining an electron from other atoms, making them negatively-charged ions. When an atom loses an electron to become a positively-charged ion, this is indicated by a plus sign after the element symbol for example, Na +. As a result of losing a negatively-charged electron, they become positively-charged ions. This means that they can achieve a stable configuration and a filled outer shell by donating or losing an electron. 
In comparison, the group 1 elements, including hydrogen (H), lithium (Li), and sodium (Na), all have one electron in their outermost shells. Their non-reactivity has resulted in their being named the inert gases (or noble gases). As shown in, the group 18 atoms helium (He), neon (Ne), and argon (Ar) all have filled outer electron shells, making it unnecessary for them to gain or lose electrons to attain stability they are highly stable as single atoms. The periodic table is arranged in columns and rows based on the number of electrons and where these electrons are located, providing a tool to understand how electrons are distributed in the outer shell of an atom. Elements in other groups have partially-filled valence shells and gain or lose electrons to achieve a stable electron configuration.Īn atom may gain or lose electrons to achieve a full valence shell, the most stable electron configuration. A full valence shell is the most stable electron configuration. Group 18 elements (helium, neon, and argon are shown) have a full outer, or valence, shell. \):īohr diagrams indicate how many electrons fill each principal shell.
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
